Transferable ionic parameters for first-principles Poisson-Boltzmann solvation calculations: Neutral solutes in aqueous monovalent salt solutions
Abstract
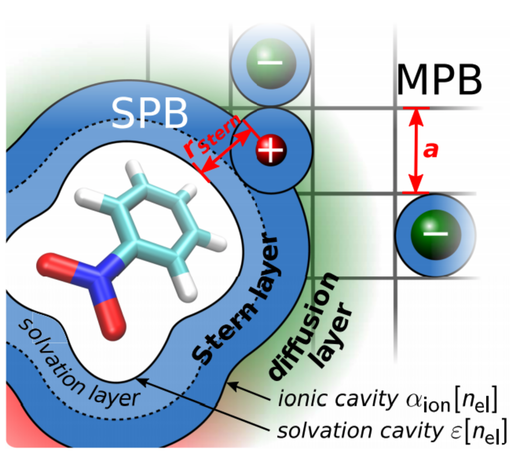
Implicit solvation calculations based on a Stern-layer corrected size-modified Poisson-Boltzmann (SMPB) model are an effective approach to capture electrolytic effects in first-principles electronic structure calculations. For a given salt solution, they require a range of ion-specific parameters, which describe the size of the dissolved ions as well as thickness and shape of the Stern layer. Out of this defined parameter space, we show that the Stern layer thickness expressed in terms of the solute’s electron density and the resulting ionic cavity volume completely determine ion effects on the stability of neutral solutes. Using the efficient SMPB functionality of the full-potential density-functional theory package FHI-aims, we derive optimized such Stern layer parameters for neutral solutes in various aqueous monovalent electrolytes. The parametrization protocol relies on fitting to reference Setschenow coefficients that describe solvation free energy changes with ionic strength at low to medium concentrations. The availability of such data for NaCl solutions yields a highly predictive SMPB model that allows to recover the measured Setschenow coefficients with an accuracy that is comparable to prevalent quantitative regression models. Correspondingly derived SMPB parameters for other salts suffer from a much scarcer experimental data base but lead to Stern layer properties that follow a physically reasonable trend with ionic hydration numbers.