Atomic origins of electrochemical stability in acetate-based dual-cation water-in-salt electrolytes
Abstract
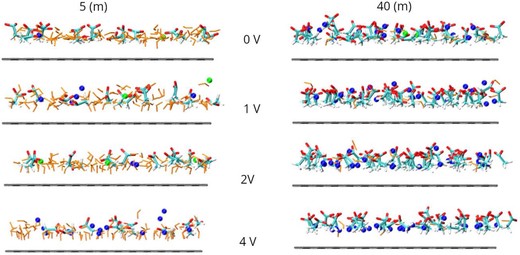
A recent study reported a super-concentrated lithium–potassium acetate-based water-in-bisalt (WiBS) electrolyte. Notably, this system exhibits a high electrochemical stability window of ∼3 V despite the absence of a conventional solid electrolyte interphase. Intriguingly, vibrational spectroscopy revealed a systematic redshift of the O–D stretch of HOD in this electrolyte with increasing salt concentration contradicting the idea that the enhanced electrochemical stability is positively correlated with the strength of the O–H bond. To explore the atomic factors determining such extended electrochemical stability of this WiBS, we investigate the molecular distributions and water reactivity at the electrified electrode–electrolyte interface using constant potential classical molecular dynamics simulations. By identifying truly interfacial molecules, we uncover a diminishing surface concentration of water molecules at the electrodes, which likely leads to a delayed onset of electrochemical processes. Furthermore, the reduced magnitude of the perturbing electric field along the reaction coordinates for electrochemical water splitting provides an atomic-level explanation for the enhanced electrochemical stability observed in this dual-cation system.