On the importance of the electric double layer structure in aqueous electrocatalysis
Abstract
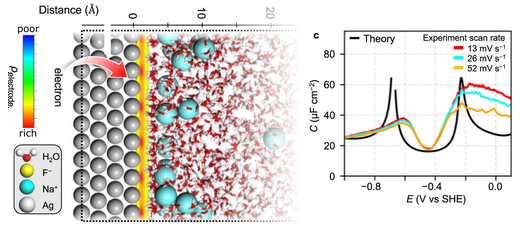
To design electrochemical interfaces for efficient electric-chemical energy interconversion, it is critical to reveal the electric double layer (EDL) structure and relate it with electrochemical activity; nonetheless, this has been a long-standing challenge. Of particular, no molecular-level theories have fully explained the characteristic two peaks arising in the potential-dependence of the EDL capacitance, which is sensitively dependent on the EDL structure. We herein demonstrate that our first-principles-based molecular simulation reproduces the experimental capacitance peaks. The origin of two peaks emerging at anodic and cathodic potentials is unveiled to be an electrosorption of ions and a structural phase transition, respectively. We further find a cation complexation gradually modifies the EDL structure and the field strength, which linearly scales the carbon dioxide reduction activity. This study deciphers the complex structural response of the EDL and highlights its catalytic importance, which bridges the mechanistic gap between the EDL structure and electrocatalysis.