Heterogeneous Catalyst as a Functional Substrate Governing the Shape of Electrochemical Precipitates in Oxygen-Fueled Rechargeable Batteries
Abstract
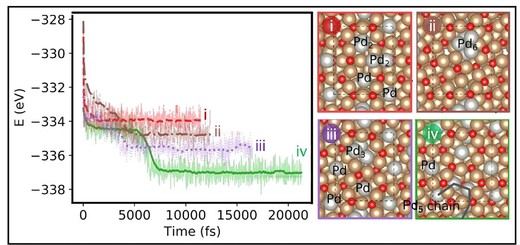
Lithium−oxygen batteries have the potential to become the most eminent solution for future energy storage with their theoretical energy density exceeding all existing batteries. However, the insulating and insoluble discharge product (lithium peroxide; Li2O2) impairs practical application. Conventional catalyst designs based on the electronic structure and interfacial charge transfer descriptors have not been able to overcome these limitations due to Li2O2. Herein, we revisit the role of heterogeneous catalysts as substrates to regulate Li2O2 growth and the formation of solid/solid reaction interfaces. We demonstrate that controlled solid/solid interfacial structure design is a critical performance parameter beyond the inherent electronic structure. In particular, the Cu2O substrate in this study induces a homogeneous deposition of Pd atoms, which leads to well-controlled growth of Li2O2 resolving mass and charge transport limits (i.e., the bottleneck of oxygen reduction/evolution reactions), thus improving reversibility, capacity, and durability of the cells by dissipating electrochemical and mechanical stress. We thus verified the essential role of solid/solid interfaces to regulate the nucleation and growth process of Li2O2 in lithium−oxygen batteries.