A unifying mechanism for cation effect modulating C1 and C2 productions from CO2 electroreduction
Abstract
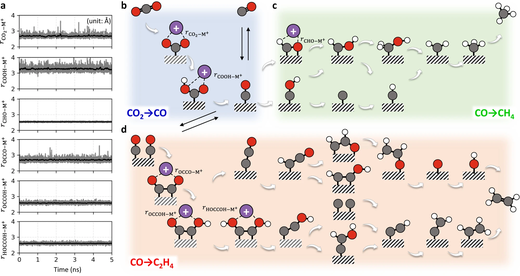
Electrocatalysis, whose reaction venue locates at the catalyst–electrolyte interface, is controlled by the electron transfer across the electric double layer, envisaging a mechanistic link between the electron transfer rate and the electric double layer structure. A fine example is in the CO2 reduction reaction, of which rate shows a strong dependence on the alkali metal cation (M+) identity, but there is yet to be a unified molecular picture for that. Using quantum-mechanics-based atom-scale simulation, we herein scrutinize the M+-coupling capability to possible intermediates, and establish H+ and M+ associated ET mechanisms for CH4 and CO/C2H4 formations, respectively. These theoretical scenarios are successfully underpinned by Nernstian shifts of polarization curves with the H+ or M+ concentrations and the first-order kinetics of CO/C2H4 formation on the electrode surface charge density. Our finding further rationalizes the merit of using Nafion-coated electrode for enhanced C2 production in terms of enhanced surface charge density.