Influence of Atomic Surface Structure on the Activity of Ag for the Electrochemical Reduction of CO2 to CO
Abstract
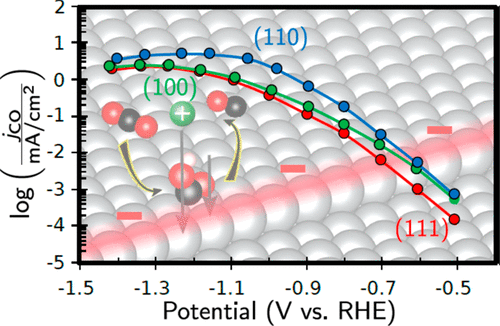
The present work was undertaken to elucidate the facet-dependent activity of Ag for the electrochemical reduction of CO2 to CO. To this end, CO2 reduction was investigated over Ag thin films with (111), (100), and (110) orientations prepared via epitaxial growth on single-crystal Si wafers with the same crystallographic orientations. This preparation technique yielded larger area electrodes than can be achieved using single-crystals, which enabled the electrocatalytic activity of the corresponding Ag surfaces to be quantified in the Tafel regime. The Ag(110) thin films exhibited higher CO evolution activity compared to the Ag(111) and Ag(100) thin films, consistent with previous single-crystal studies. Density functional theory calculations suggest that CO2 reduction to CO is strongly facet-dependent, and that steps are more active than highly coordinated terraces. This is the result of both a higher binding energy of the key intermediate COOH as well as an enhanced double-layer electric field stabilization over undercoordinated surface atoms located at step edge defects. As a consequence, step edge defects likely dominate the CO2 reduction activity observed over the Ag(111) and Ag(100) thin films. The higher activity observed over the Ag(110) thin film is then related to the larger density of undercoordinated sites compared to the Ag(111) and Ag(100) thin films. Our conclusion that undercoordinated sites dominate the CO2 reduction activity observed over close-packed surfaces highlights the need to consider the contribution of such defects in studies of single-crystal electrodes.